Resources
SUPPORT EVERY STEP OF THE WAY
Explore the multiple resources available to help you stay informed and feel supported every step of the way.
Getting Started Is Easy

Once you or your child is prescribed LIVMARLI, Mirum Access Plus experts will help you navigate your insurance coverage and share financial support options that may help lower out-of-pocket costs.
Mirum Access Plus Savings Program
If you have commercial insurance and your insurance coverage for LIVMARLI is approved:
Eligible patients may pay as little as $10 out of pocket per fill for LIVMARLI‡
Mirum Patient Assistance Program (PAP)
If you are uninsured or coverage is denied, the Mirum Access Plus team will determine your eligibility for the Mirum PAP. Mirum PAP allows you to:
Receive LIVMARLI at no cost to you for up to 1 calendar year, with eligibility reviewed each year†
*Data from 1/1/2024 to 12/31/2024.
†Exceptions apply for some patients, including those with TRICARE insurance.
‡Subject to program terms and conditions.
Mirum Access Plus Resources

Mirum Access Plus Downloadable Resources
Mirum Access Plus is here to provide you with the resources and education you need, when you need them. Whether it’s answers to your questions, assistance with insurance coverage, or helping you refill your medication, Mirum Access Plus makes it easy to get what you need.
Mirum Access Plus Mobile
Designed for busy families on the go, Mirum Access Plus Mobile is a convenient text messaging service that makes communicating with Mirum Access Plus even easier.

If you have any questions about Mirum Access Plus, contact us at:
1-855-MRM-4YOU (1-855-676-4968) Monday through Friday, 8:00 AM through 8:00 PM ET.
Talking to Your Doctor
Understanding Alagille syndrome and its treatment can be difficult. Speaking with your child’s doctor can help make things easier.
When partnering with your doctor, conversation is key. It’s helpful to be open and honest so you can work as a team towards your treatment goals. Here are a few things to get the conversation started:
Tell your doctor
- How the itch impacts daily life
- Treatment history
- Any previous surgeries or transplants
- Other conditions you or your child have
- Medicines you or your child take
- Specific goals related to health, treatment, and management of Alagille syndrome
Ask your doctor
- Could LIVMARLI be an option for me or my child?
- How will I know if LIVMARLI is working?
- How is LIVMARLI taken?
- What are the possible side effects? How can they be managed?
- Can LIVMARLI be taken if someone has other health conditions or takes other medicines?
- What support is available while taking LIVMARLI?
- How do we get started?
Support at Your Fingertips

The Alagille Syndrome Alliance (ALGSA)
ALGSA is a non-profit organization with a goal of supporting the Alagille syndrome community by mobilizing resources, facilitating connections, promoting unity, and advocating for a cure to inspire, empower, and enrich the lives of people affected by Alagille syndrome.

Yellow Alert is the Children’s Liver Disease Foundation’s awareness campaign promoting the early identification of liver disease in infants and their referral.

CLASS is an all-volunteer group helping children affected with liver diseases by addressing the emotional, educational, and financial needs of families.

The National Organization for Rare Disorders (NORD) is a patient advocacy organization dedicated to individuals with rare diseases and the organizations that serve them. NORD is committed to the identification, treatment, and cure of rare disorders through programs of education, advocacy, research, and patient services.

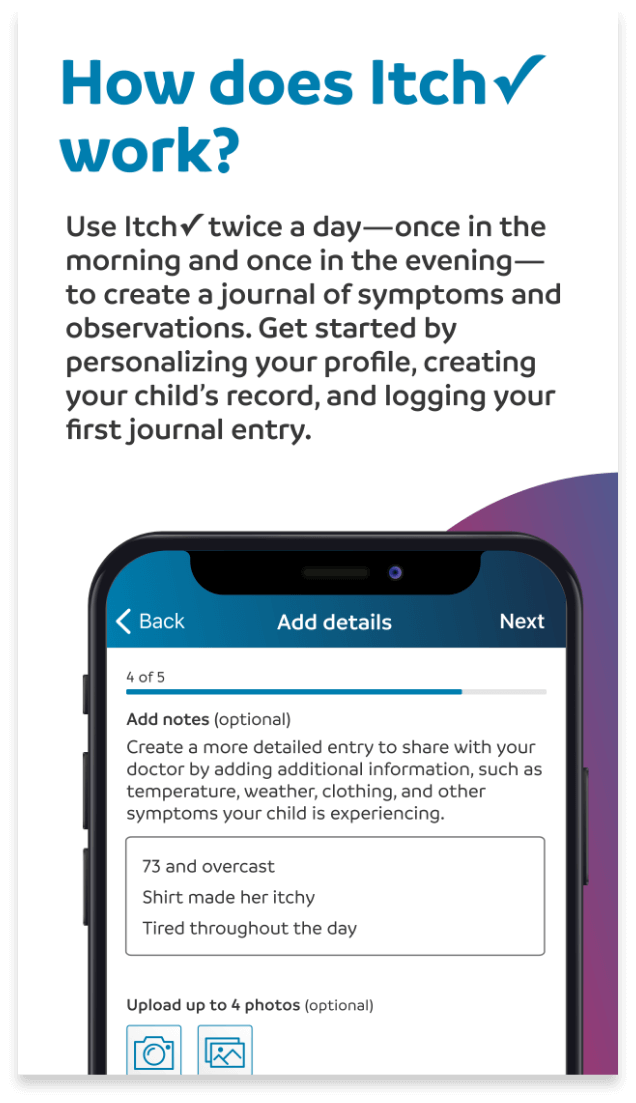
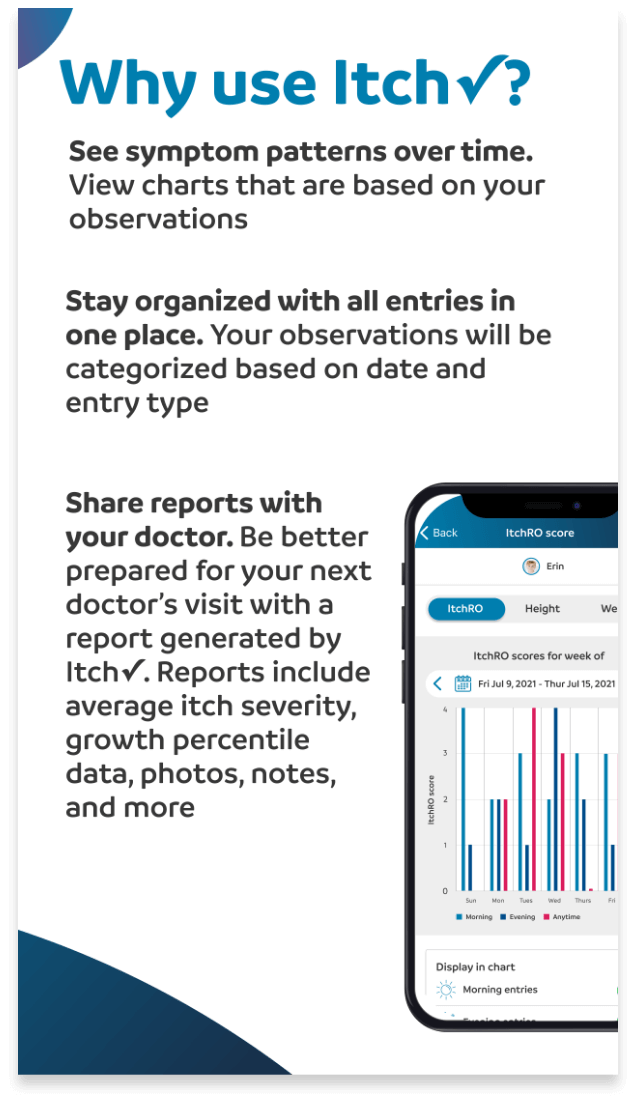
Check it.
Chart it.
Discuss It.™
Introducing the Itch✓ app: An easy-to-use journal designed to help you and your doctor keep track of your itch.