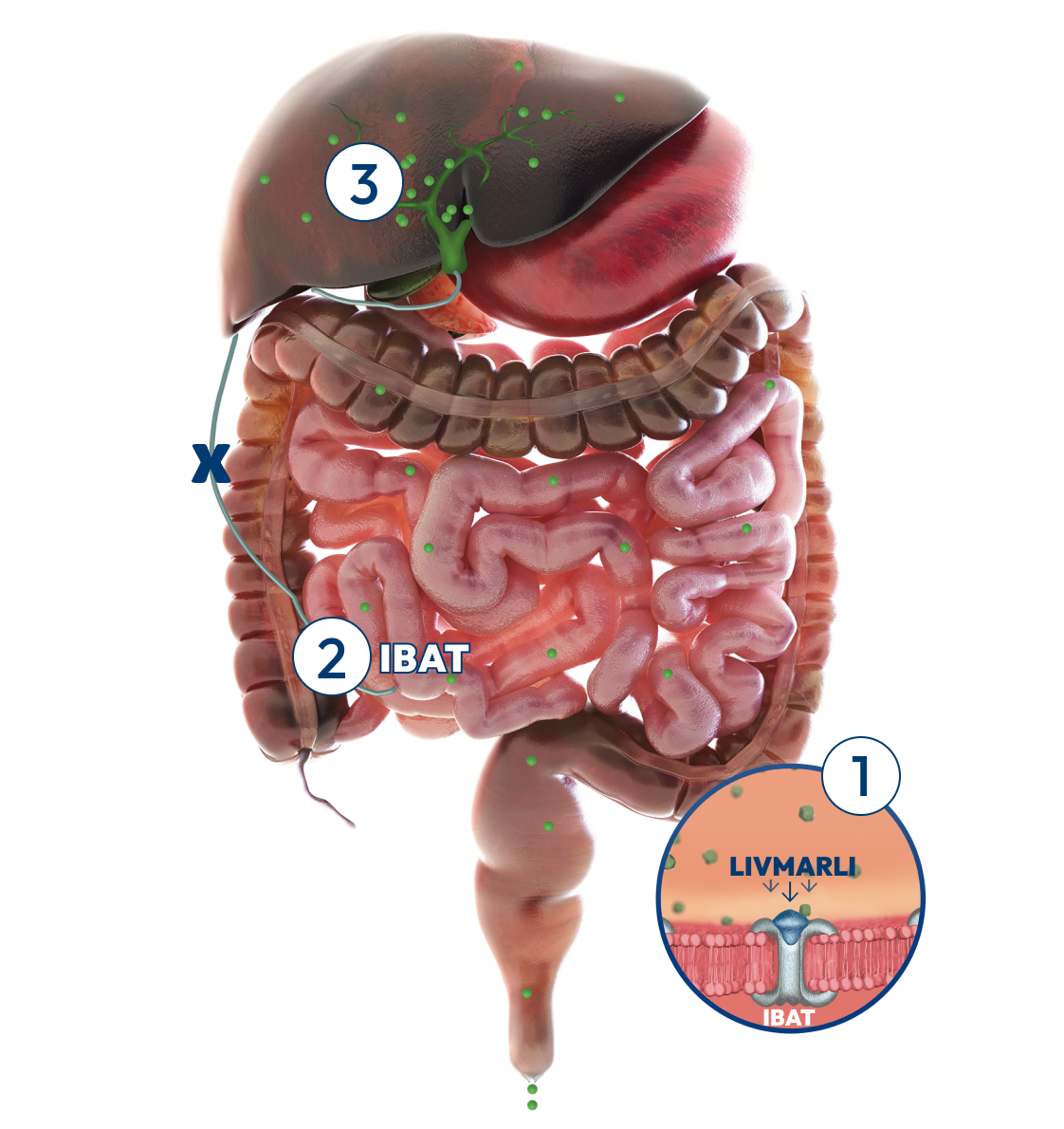
What are the possible side effects of LIVMARLI (maralixibat) oral solution and tablets?
LIVMARLI can cause serious side effects, including:
- Liver injury. Changes in certain liver tests are common in patients with PFIC but may worsen during treatment. These changes may be a sign of liver injury and can be serious or may lead to liver transplant or death. Your health care provider should do blood tests and physical exams before starting and during treatment with LIVMARLI to check your liver function. Tell your health care provider right away if you get any signs or symptoms of liver problems, including:
- nausea or vomiting
- your skin or the white part of your eye turns yellow
- dark or brown urine
- pain on the right side of your stomach (abdomen)
- fullness, bloating, or fluid in your stomach area (ascites)
- loss of appetite
- bleeding or bruising more easily than normal, including vomiting blood
Stomach and intestinal (gastrointestinal) problems. LIVMARLI can cause stomach and intestinal problems, including diarrhea and stomach pain during treatment. Diarrhea can also cause the loss of too much body fluid (severe dehydration). Your health care provider may advise you to monitor for new or worsening stomach problems, including stomach pain, diarrhea, blood in your stool, or vomiting
Tell your health care provider right away if you have any new or worsening signs or symptoms of stomach and intestinal problems, including:
- diarrhea
- more frequent bowel movements than usual
- stools that are black, tarry, or sticky, or have blood or mucous
- severe stomach-area pain or tenderness
- vomiting
- urinating less often than usual
- dizziness
- headache
- A condition called Fat-Soluble Vitamin (FSV) Deficiency caused by low levels of certain vitamins (vitamin A, D, E, and K) stored in body fat. FSV deficiency is common in patients with PFIC but may worsen during treatment. Your health care provider should do blood tests before starting and during treatment, and may monitor for bone fractures and bleeding, which are common side effects
Tell your health care provider about all medicines that you take, as LIVMARLI may interact with other medicines. If you take a medicine that lowers cholesterol by binding bile acids, such as cholestyramine, colesevelam, or colestipol, take LIVMARLI at least 4 hours before or 4 hours after you take that medicine.
Your health care provider may change your dose, or temporarily or permanently stop treatment with LIVMARLI if you have certain side effects.
LIVMARLI is available in oral solution and tablet formulations. Take LIVMARLI exactly as your health care provider tells you to and do not change your dose unless you are instructed to. In patients with PFIC, LIVMARLI is taken by mouth, 2 times each day, 30 minutes before a meal. For the oral solution, be sure to use the provided oral dosing dispenser to accurately measure the dose of medicine.
These are not all of the possible side effects of LIVMARLI. For more information, ask your health care provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
Please see full Prescribing Information, including Patient Information and Instructions for Use, for LIVMARLI.